3D Printed Adapters for Decathlon Snorkel Mask Conversion to Emergency PPE
What are Snorkel Mask to Emergency PPE Adapters?
Problem
Front-line providers are running out of personal protective equipment (PPE). At many hospitals, there is a limited, or total lack of supply of PPE, which leaves healthcare professionals without the necessary resources to perform their job, raising their chances of infection.
How are the Mask Adapters Made?
Solution
Adapting snorkel masks with connectors makes it possible to include filters in the entry/exit of the mask to filter the air of both the user and surroundings, while isolating the whole face. The hospitals involved in this project have reported that the masks can be reused, as they can be disinfected and/or sterilized according to each hospital's materials and protocols, which is a key advantage compared to traditional masks.
SLA 3D printing technology and materials have been selected to produce the models due to their durability, rapid fabrication, and scalability at the industrial level.
From the materials, Formlabs Draft Resin was chosen for this project as it can be used to manufacture adapters up to four times faster compared to Standard Resins. With Draft Resin, the production capacity of a single Form 3 or Form 3B SLA 3D printer is around 40-50 units/day.
Note: Formlabs does not endorse using Draft Resin for this as it has not been evaluated as safe for this application. See the disclaimer for more information.
“Seeing the evolution of the pandemic and news in Italy, we knew it was a matter of time that new opportunities and high impact solutions came up for hospitals using Formlabs SLA 3D printing. For this reason, we created a multi-company working team that could provide an integral solution: design, prototyping, fabrication, and delivery to hospitals. At 3Digital Factory, we already had extensive experience in the dental-healthcare sector and had access to 7 Formlabs printers in our facilities at 3D Factory Incubator. We could move an extensive web of printers across the country with the help of Formlabs and SolidPerfil 3D. A few days after, we started to receive plenty of helping requests and since that moment we managed to deliver more than 800 parts to convert the Decathlon snorkel face mask into an emergency PPE. We are now in the development and validation phases of a printed part that would allow us to convert this same mask into a non-invasive PPV mask for patients.” - Alan Alves, 3Digital Factory CEO.
Validation Status
The Hospital Clínico Universitario de Valladolid and the Hospital Universitario Infanta Leonor have used the connectors with positive results. The printed part fits perfectly and no leakages are detected.
The printed parts were also tested by the hospitals involved to withstand disinfection and sterilization procedures that are compatible with the snorkel mask, including enzymatic detergent and being washed*, dried in the machine at 60ºC* temperature and hydrogen peroxide gas plasma sterilization (50ºC)*.
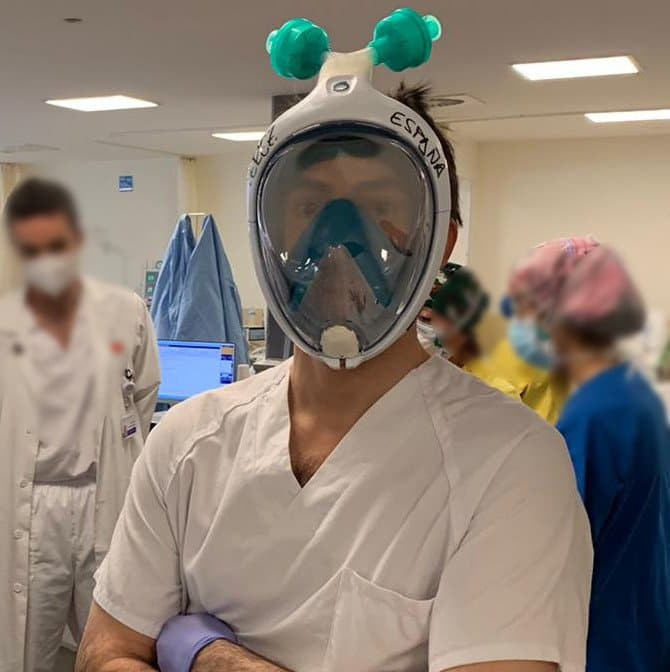
Dr. Manuel Pérez-España from Hospital Universitario Infanta Leonor (Madrid)
“The big advantage for us is the possibility to re-sterilize the same part several times, the quality and reproducibility of the printers together with the obtained sealing” - Dr. Alfredo Redondo, Hospital Clínico Universitario de Valladolid

Dr. Raúl del Castillo wore the Decathlon Snorkel Mask adapted to PPE via a 3D printed connector for two hours with success which involved performing a tracheotomy on a patient with COVID-19. The surgery was conducted in the Hospital Universitario Quirón Pozuelo. These masks are also being used in the Hospital Universitario Ramón y Cajal where the doctor works.
The printed parts were also tested by the hospitals involved to withstand disinfection and sterilization procedures that are compatible with the snorkel mask, including enzymatic detergent and being washed, dried in the machine at 60ºC temperature and hydrogen peroxide plasma sterilization (50ºC).
Hospitals involved in the validation process:
-
Hospital Clínico Universitario de Valladolid
-
Hospital Universitario Ramón y Cajal (Madrid)
-
Hospital Universitario Infanta Leonor (Madrid)
-
Hospital de Igualada
How to Support the Cause
3Digital Factory, is printing free of charge devices to help hospitals in need of an emergency PPE in the COVID-19 crisis.
We're working with the hospitals and 3Digital Factory to make the digital files as well as printing and assembly instructions available to the public in the near future. For more information, reach out to 3Digital Factory.
Disclaimer
*It is the Hospital's responsibility to perform disinfection procedures on a daily basis and confirm the quality of the mask and printed parts after undergoing disinfection and/or sterilization procedures. There are no clinical studies regarding the potential degradation of the elements over time.
Formlabs has not independently verified or validated the parts mentioned in this case study and if they are safe or effective for their intended purpose. Formlabs resins have not been evaluated for resin VOC for any specific application, standard, or occupational hazard. Analytical data suggests Surgical Guide VOC under airflow or oxygen flow is in the range of ~100 ppb.
Any 3D printed appliances that are subjected to skin contact should meet the biocompatibility requirements stated in ISO 10993-1. Please note that Formlabs materials not categorized as biocompatible, including Draft Resin, were not designed to meet these requirements. If your specific performance needs are beyond the scope of the original non-biocompatible material design, please consult Formlabs for further information on the potential use of secondary processing.
Any additional modifications outside the published guidance for use must be tested by the manufacturer prior to implementation.
Page Last Updated: April 6, 2020

