
- Print Technology
- SLA
- Colors
- Translucent
- Characteristics
- Biocompatibility, Elastomeric, Hard Rubber-Like
- Ultimate Tensile Strength
- 7.2 MPa
- Stress at 100% Elongation
- 4.5 MPa
- Elongation at Break
- 135.0 %
- Tear Strength
- 22.0 kN/m
- Shore Hardness
- 80.0 A
- Compression Set 70°C for 22 hours
- 5.3 %
BioMed Flex 80A Resin is a firm, flexible, and transparent material for biocompatible applications requiring durability and long-term skin (>30 days) or short-term mucosal membrane contact (<24 hours).
BioMed Flex 80A Resin is manufactured in our FDA-registered, ISO 13485-certified facility.
- Print Technology
- SLA
- Colors
- Translucent
- Characteristics
- Biocompatibility, Elastomeric, Hard Rubber-Like
- Ultimate Tensile Strength
- 7.2 MPa
- Stress at 100% Elongation
- 4.5 MPa
- Elongation at Break
- 135.0 %
- Tear Strength
- 22.0 kN/m
- Shore Hardness
- 80.0 A
- Compression Set 70°C for 22 hours
- 5.3 %
Select Printer Series and Resin Volume
Bulk Order Discount
- 15%
- 30 L
- 20%
- 60 L
- 25%
- 120 L
- 30%
- 240 L
- 35%
- 1,000 L
- 40%
- 2,000 L
- 45%
- 3,000 L
Compatibility
- 3D Printer
- Form 4BL, Form 4B, Form 3B, Form 3BL
- Build Platform
- Form 4 Build Platform, Form 4 Flex Build Platform, Form 3 Build Platform, Form 3 Build Platform 2, Form 3 Stainless Steel Build Platform, Form 3L Build Platform, Form 3L Build Platform 2L, Form 4L Build Platform, Form 4L Flex Build Platform
- Tank
- Form 3 Resin Tank V2.1, Form 3L Resin Tank V3, Form 4 Resin Tank, Form 4L Resin Tank
BioMed Flex 80A Resin—
Why Choose BioMed Flex 80A Resin?
Achieve new design possibilities and efficiencies using 3D printing and BioMed Flex 80A Resin, from producing flexible biocompatible medical devices to anatomical models requiring biocompatibility.
Flexible
Produce flexible medical devices or firm tissue models requiring short-term mucosal membrane contact that can optimize mass personalization applications versus using cumbersome molds.
Transparent
Produce complex transparent firm tissue models and bring them directly into the operating room (O.R.) as a reference for complex medical procedures (eg. sizing models, cartilage models, or disease organs.)
Medical-Grade Material, Biocompatible Parts
Leverage a material manufactured within our Quality Management Systems, with strict adherence to ISO 13485. Instill confidence that this material is suitable for biocompatible applications with long-term skin (>30 days), short-term mucosal membrane contact (<24 hours), and USP Class VI certifications.
Applications*
Directly 3D print flexible parts requiring biocompatibility with BioMed Flex 80A Resin. Reduce workflow times by eliminating molding to directly produce flexible, patient-specific medical devices or firm tissue medical models that surgeons can reference in the O.R.
*Unlike Formlabs Dental Resins, no intended uses are provided for BioMed Resins. Material properties may vary based on part design, manufacturing practices, and other methods. It is the manufacturer’s responsibility and its respective customers and end-users to determine the biocompatibility and performance of all printed parts for their respective application and use. To instill confidence in potential users, Formlabs has tested biocompatibility for common use cases and manufactures BioMed Resins in an ISO 13485-certified facility. We can offer representative examples of what customers have done with our products and the supporting documentation we make freely available.
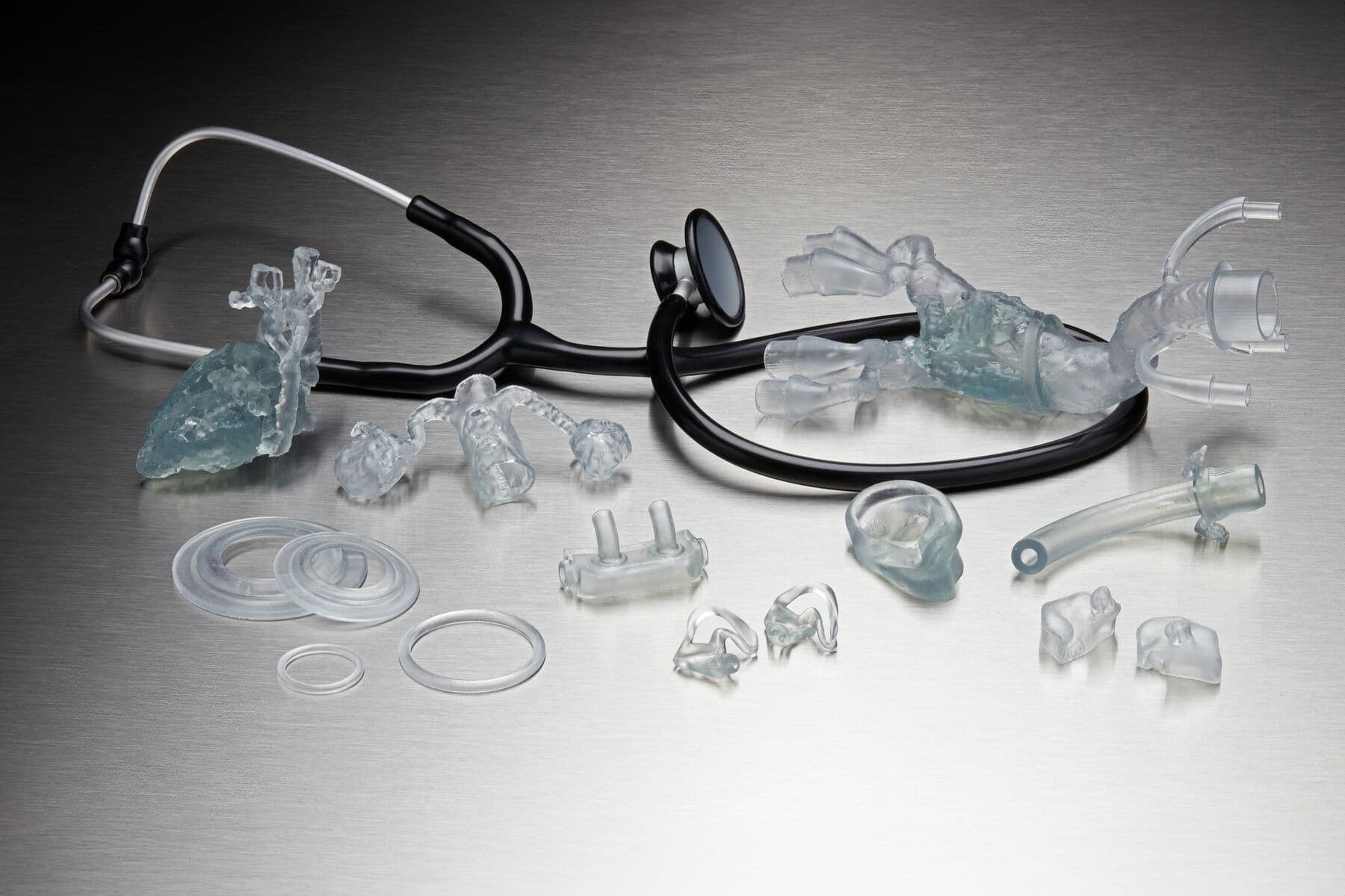
Flexible biocompatible medical devices and components, and firm tissue models to assist in surgeries

Medical devices requiring short-term mucosal membrane contact

Manufactured in an ISO 13485-Certified Facility
Formlabs manufactures our Biomed and Medical Device Resins at our FDA-registered facility in Ohio. These materials are designed and manufactured within our robust Quality Management System that is ISO 13485 and EU MDR certified. A dedicated team of operators and quality assurance professionals makes the resins inside a certified ISO Class 8 clean room. All of our Medical Device Resins are appropriately registered with the FDA and CE marked according to the EU MDR.
Material Properties*
| Parameter |
Value |
|---|---|
|
Ultimate Tensile Strength |
7.2 MPa |
|
Stress at 50% Elongation |
2.6 MPa |
|
Stress at 100% Elongation |
4.5 MPa |
|
Elongation |
135% |
|
Tear Strength |
22 kN/m |
|
Shore Hardness |
80 A |
Post-Processing
Washing
The foundational step in any SLA post-processing workflow is to remove excess resin on the surface of the parts through an alcohol wash. Please refer to the material's manufacturing guide/IFU for washing instructions.
SETTINGS
Curing
For biocompatible materials, post-curing is necessary to achieve the safety standards determined by regulatory agencies. Please follow the material’s manufacturing guide/IFU for proper post-processing.
SETTINGS