- Print Technology
- SLA
- Colors
- Translucent
- Applications
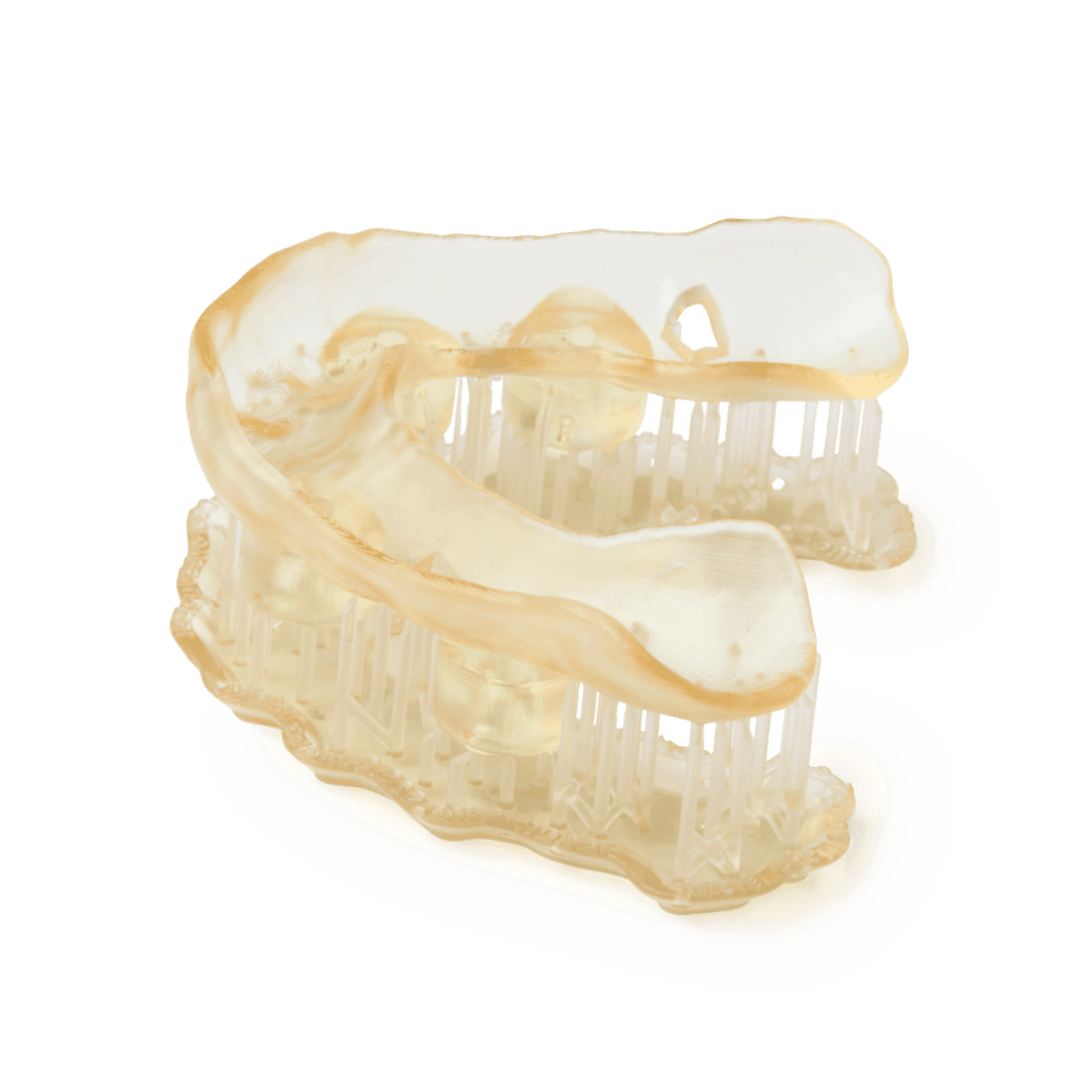
- Surgical Guides
- Characteristics
- Biocompatibility, Sterilizable
- Flexural Modulus
- 2400.0 MPa
- Elongation at Break
- 12.0 %
- Flexural Strength
- 102.0 MPa
Surgical Guide Resin is an autoclavable, biocompatible resin for 3D printing surgical guides, drilling templates, pilot drill guides, and device sizing templates that exceed dental demands in accuracy, part quality, and performance. Surgical Guide Resin was formulated specifically for Formlabs printers and rigorously tested to meet solvent disinfection and autoclave sterilization standards for implant systems.
Read our application guide to learn the complete digital workflow and best practices for making 3D printed surgical guides on your Formlabs 3D printer.
Regional Availability
Product availability is limited to certain regions. See regional availability.
- Print Technology
- SLA
- Colors
- Translucent
- Applications
- Surgical Guides
- Characteristics
- Biocompatibility, Sterilizable
- Flexural Modulus
- 2400.0 MPa
- Elongation at Break
- 12.0 %
- Flexural Strength
- 102.0 MPa
Select Printer Series and Resin Volume
Bulk Order Discount
- 15%
- 30 L
- 20%
- 60 L
- 25%
- 120 L
- 30%
- 240 L
- 35%
- 1,000 L
- 40%
- 2,000 L
- 45%
- 3,000 L
Compatibility
- 3D Printer
- Form 4BL, Form 4B, Form 3B, Form 3BL, Form 2
- Build Platform
- Form 4 Build Platform, Form 4 Flex Build Platform, Form 3 Build Platform, Form 3 Build Platform 2, Form 3 Stainless Steel Build Platform, Form 3L Build Platform, Form 3L Build Platform 2L, Form 4L Build Platform, Form 4L Flex Build Platform
- Tank
- Form 2 LT Tank, Form 3 Resin Tank V2.1, Form 3L Resin Tank V3, Form 4 Resin Tank, Form 4L Resin Tank
Surgical Guide Resin—
Why Choose Surgical Guide Resin?
Surgical Guide Resin provides dental professionals with a biocompatible material for the production of dimensionally accurate dental implant guides and templates.

Reliable
Consistency produce reliable guides.
Efficient
Leverage a digital workflow from intraoral scanning to production.
Biocompatibility
Provide the patient and clinician the peace of mind they deserve.
Applications
Surgical Guide Resin is a premium-quality material for printing surgical implant guides.
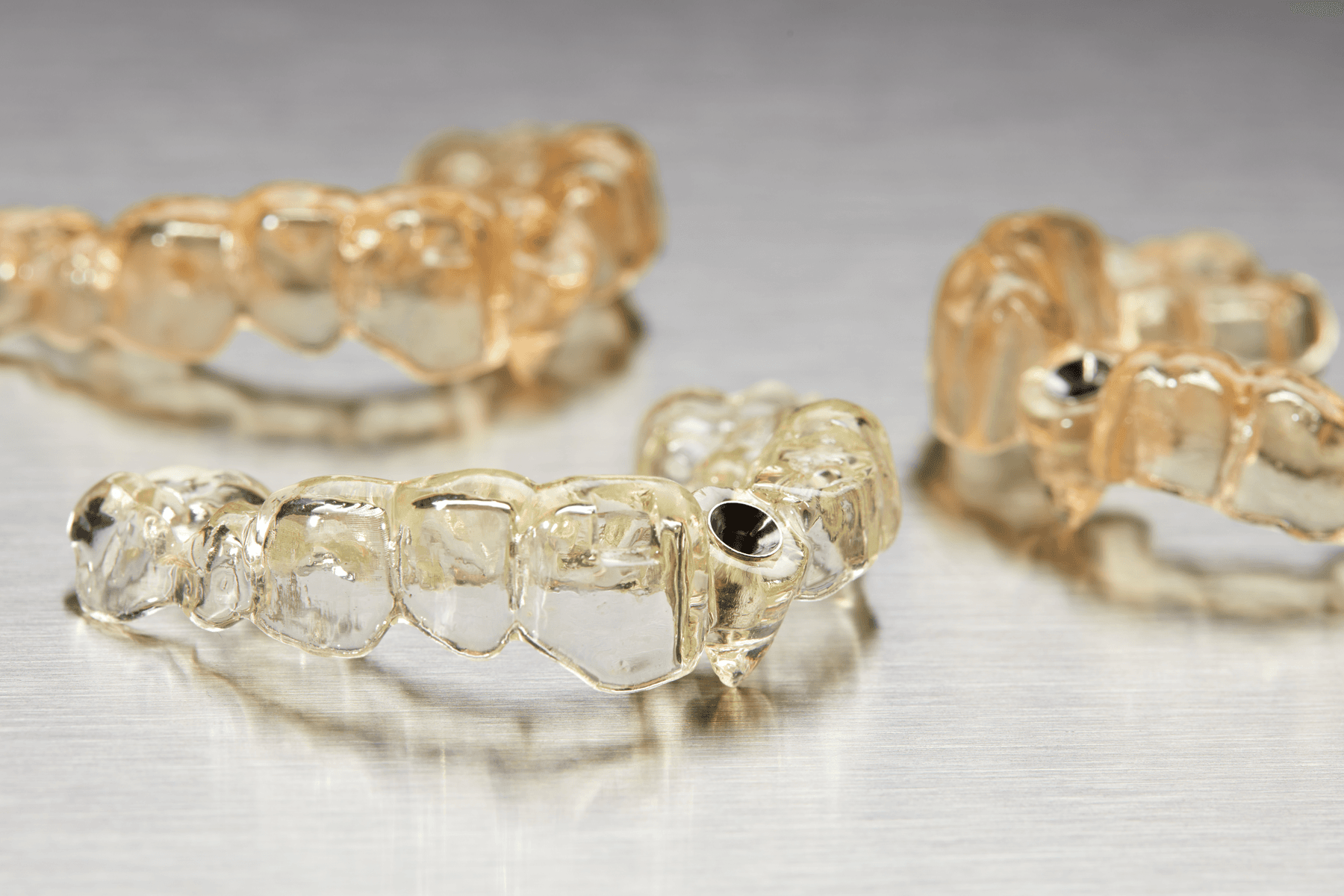
Splints
Device Sizing Templates
Occlusal Guards
Drilling Templates

Manufactured in an ISO 13485-Certified Facility
Formlabs manufactures our Biomed and Medical Device Resins at our FDA-registered facility in Ohio. These materials are designed and manufactured within our robust Quality Management System that is ISO 13485 and EU MDR certified. A dedicated team of operators and quality assurance professionals makes the resins inside a certified ISO Class 8 clean room. All of our Medical Device Resins are appropriately registered with the FDA and CE marked according to the EU MDR.
Material Properties*
| Parameter |
Value |
|---|---|
|
Elongation |
12% |
|
Flexural Strength |
> 102 MPa |
|
Flexural Modulus |
> 2400 MPa |
* Material properties may vary based on part geometry, print orientation, print settings, and temperature. Data for post-cured samples were measured on Type IV tensile bars printed on a Form 2 printer with 100 μm Surgical Guide Resin settings, washed in a Form Wash for 20 minutes in ≥99% isopropyl alcohol, and post-cured at 60 °C for 30 minutes in a Form Cure.
Post-Processing
Washing
Surgical Guide Resin may be washed using either a Formlabs-validated wash unit (1) or an ultrasonic wash unit (2).
The foundational step in any SLA post-processing workflow is to remove excess resin on the surface of the parts through an alcohol wash. Please refer to the material's manufacturing guide/IFU for washing instructions.
SETTINGS
Curing
For biocompatible materials, post-curing is necessary to achieve the safety standards determined by regulatory agencies. Please follow the material’s manufacturing guide/IFU for proper post-processing.
SETTINGS
Request a Free Surgical Guide Resin Sample Part
