3D-Druck zur Unterstützung der COVID-19 Response
Formlabs möchte der medizinischen Fachwelt helfen, 3D-Druck zur Bekämpfung der COVID-19-Pandemie und der damit verbundenen Lieferkettenengpässe zu nutzen. Wir kooperieren mit Dutzenden von Krankenhäusern, Gesundheitssystemen und staatlichen Stellen auf der ganzen Welt bei verschiedenen Projekten, von COVID-19-Tests, über PSA bis zu medizinischem Gerät.
Wenn Sie eine Organisation im Gesundheitswesen sind, die überlegt, 3D-Druck für COVID-19 oder andere medizinische Anwendungsfälle zu nutzen, sollten Sie unseren Leitfaden: Überlegungen für die Herstellung sicherer und effektiver Medizinprodukte lesen.

Medizin
Anwendungen für Krankenhäuser, Firmen für Medizinprodukte, Anbieter im Gesundheitswesen und Kliniken.
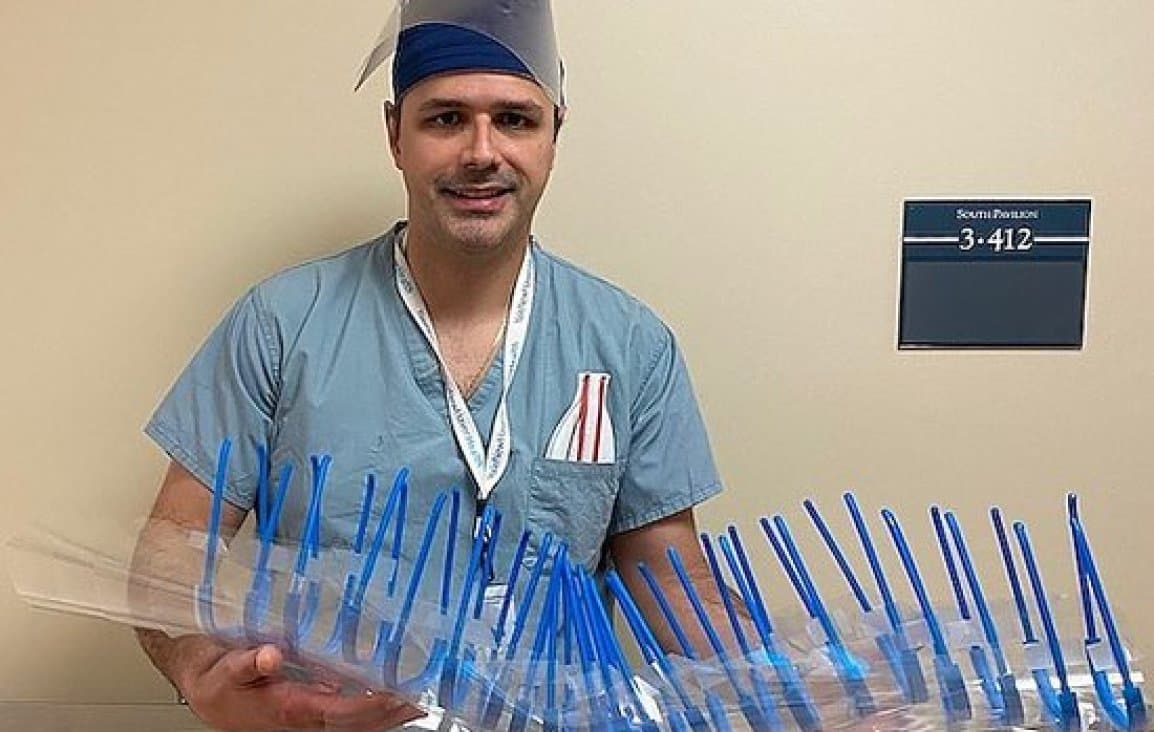
Community
Nicht klinisches Zubehör kann von jedem gedruckt und verwendet werden.
Der ultimative Leitfaden zum Maschinenbau von zu Hause
Mit der korrekten Einrichtung und dem passenden Arbeitsprozess halten Sie mit 3D-Druck für den Desktop auch zuhause die Produktivität aufrecht und passen sich schnell an. Mit dieser Agilität bleiben Sie auch in der Krise Ihren Konkurrenten voraus und halten knappe Fristen ein.

In den News
Unsere Nutzer überraschen uns immer wieder mit innovativen und bahnbrechenden 3D-Druckanwendungen für ihre Branchen, und das Gesundheitswesen bildet hierbei keine Ausnahme. Mehrere Formlabs-Kunden sorgen bereits für Schlagzeilen damit, wie sie Formlabs' Produkte im Kampf gegen COVID-19 einsetzen.
Häufig gestellte Fragen
Formlabs hat Tausende von Anfragen dazu erhalten, wie 3D-Druck in der aktuellen Krise helfen kann. Hier sind die Antworten auf einige der häufigsten Fragen, die wir erhalten haben.
Abonnieren Sie unseren monatlichen Newsletter, um auf dem Laufenden darüber zu bleiben, wie 3D-Druck die Gesundheitsbranche transformiert.
Neu beim 3D-Druck? Erkunden Sie unsere Ressourcen für das Gesundheitswesen, um zu erfahren, wie 3D-Druck dazu beiträgt, Behandlungen und Produkte zu liefern, die individuell auf jeden einzelnen Patienten zugeschnitten sind, und Zeit und Kosten spart, vom Labor bis in den Operationssaal.
Häufig gestellte Fragen
Formlabs hat Tausende von Anfragen dazu erhalten, wie 3D-Druck in der aktuellen Krise helfen kann. Hier sind die Antworten auf einige der häufigsten Fragen, die wir erhalten haben.








